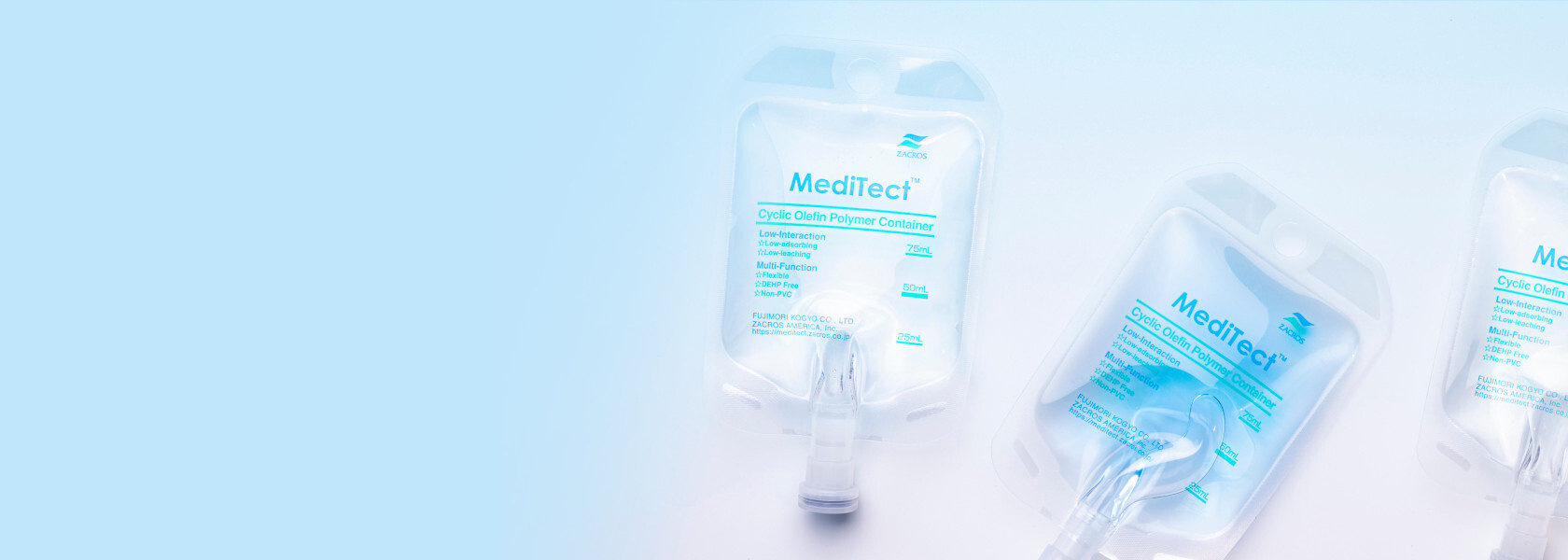
Our Product Lineup
About our MediTect™ series
The purpose and design of the MediTect™ series is to give ultimate stability to more sensitive drug contents. Typically, injectable drugs known to be unstable in plastic are packaged in glass bottles, vials, etc.
Our aim with MediTect™ is to replace glass with more “flexible” solutions!

Packaging system options
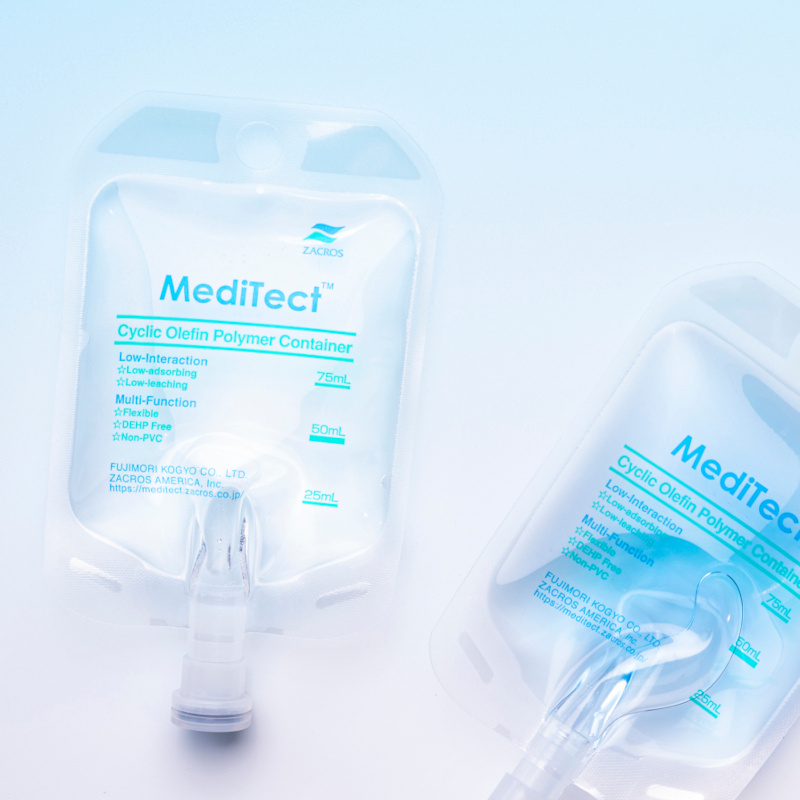
Our IV bags have many different construction options. PE and PP blends are available for different sterilization methods. Closures can be either rubber infusion caps, or even "One-touch caps" assuming aseptic filling. For additional barrier properties, overwraps are also part of our total solution for your pharmaceuticals.
Technology & Benefits

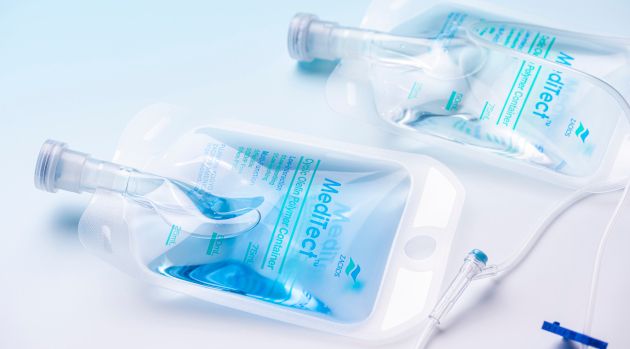
NI, standing for non-interactive, emphasizes the very purpose of our bags.
- NI films prevent elution and sorption, demonstrating glass-like storage stability. These films are the right choice for sensitive liquid drugs.
- Learn more

About Us
From project inception to go-to-market, we are with you every step of the way

- Start-to-finish collaborative packaging assessments
- Introduction to our CMO network
ZACROS has the test machines required to generate new data on prototype packaging. From sterilization systems to filling and transport test; we can create custom packaging samples and test them according to user requirements.
Our commitment to quality

- Our primary pharmaceutical packaging manufacturing site is ISO 15378 and ISO 13485 certified
- Our bags are compliant with USP and JP standards
We have sold primary packaging for parenteral drugs to the US and Japan for over 30 years, and some products are DMF registered.
With our experience, we are positioned to support you and your product launch.